Clusterprojekt Clean Circles
Allgemein
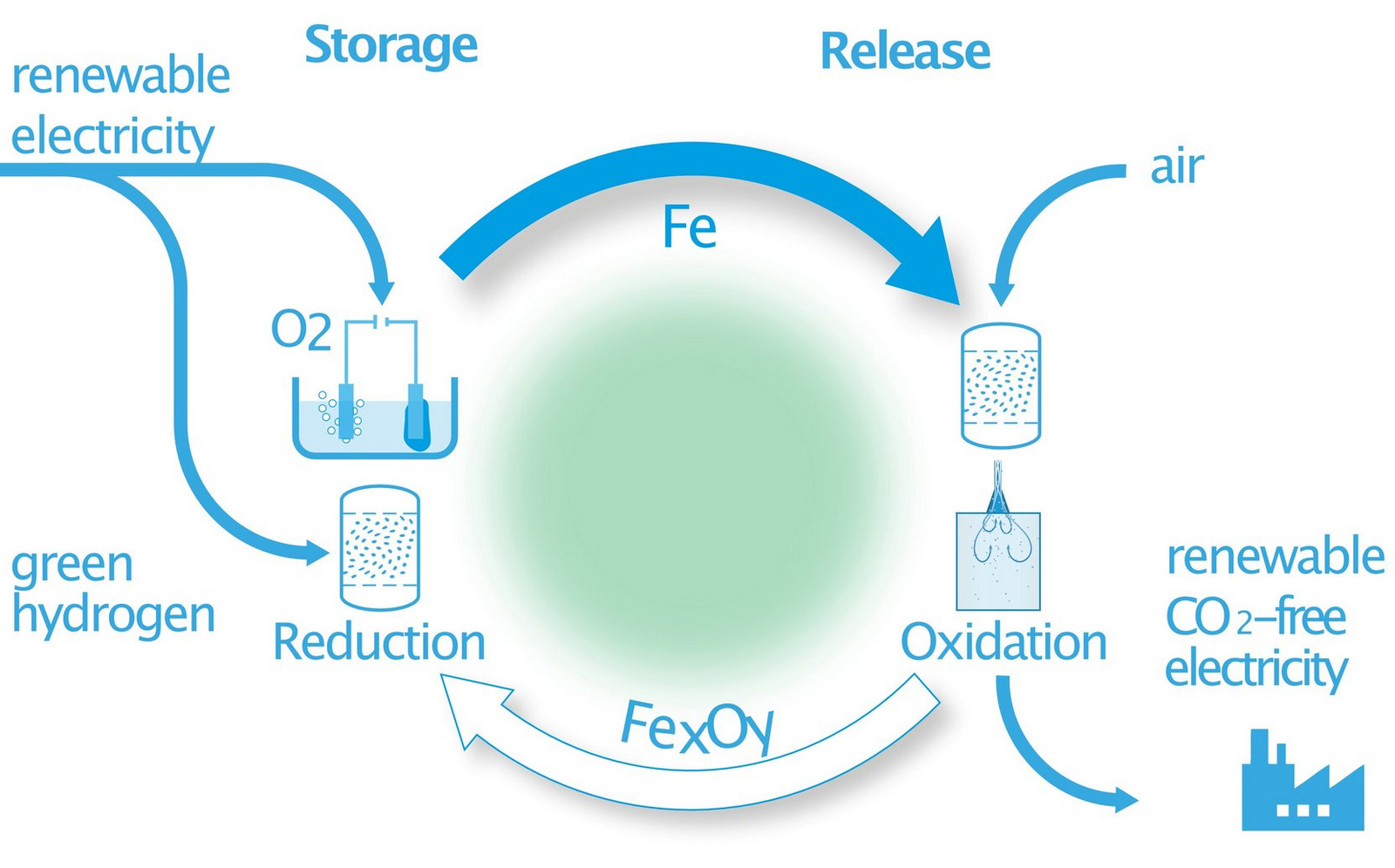
In einem einzigartigen Forschungsansatz sollen Eisen und seine Oxide in einem Kreislauf als kohlenstofffreier chemischer Energieträger für erneuerbare Energie genutzt werden (siehe Abbildung rechts). Erneuerbare Energie wird genutzt, um Eisenoxide über einen thermochemischen oder elektrochemischen Prozess zu reduzieren (Speicherung, Reduktion). Örtlich und zeitlich getrennt davon kann elementares Eisen oxidiert werden, um thermische Energie zur Stromerzeugung freizusetzen (Freisetzung, Oxidation). Auf diese Weise kann erneuerbare Energie in großen Mengen gespeichert, transportiert und CO2-frei zur Verfügung gestellt werden, eine bisher ungelöste zentrale Herausforderung der Energiewende unter sich verändernden politischen Rahmenbedingungen.
Eisen als Energieträger hat hervorragende physikalisch-chemische Eigenschaften in Bezug auf Transport, Speicherung und Energienutzung. Wind- und sonnenreiche Standorte innerhalb und außerhalb Deutschlands können in eine CO2-freie Kreislaufwirtschaft zur kostengünstigen Erzeugung und Nutzung von regenerativem Strom eingebunden werden. Für die Realisierung dieser umweltfreundlichen Lösung muss die enge und komplexe Kopplung von ingenieur-, natur-, politik- und wirtschaftswissenschaftlichen Fragestellungen berücksichtigt werden.
Für die Reduktion und Oxidation von Eisen(oxid) müssen zunächst chemische (heterogene) Reaktionen und deren Kopplung an Transportprozesse für isolierte Prozesse und einzelne Partikel verstanden werden. Reaktionsratenbestimmende Phänomene sind der Wärme- und Stoffübergang zwischen Partikeln und der umgebenden Gasphase sowie die Kinetik der heterogenen Partikeloberflächenreaktion. Diese Prozesse werden stark von den Umgebungsbedingungen (Temperatur, Zusammensetzung der Gasatmosphäre) und den Partikeleigenschaften (Partikelgröße, Phase, Porosität, etc.) beeinflusst. Dementsprechend ist eine räumlich und zeitlich aufgelöste experimentelle und numerische Analyse der Oxidations- und Reduktionsphänomene erforderlich, um das grundlegende Verständnis zu verbessern.
Arbeitspaket Prof. Dr.-Ing. Dirk Geyer
Nicht-intrusive, zeitaufgelöste Diagnostik von Mikropartikeln auf Eisenbasis, bei thermo-chemischer Reduktion oder Oxidation
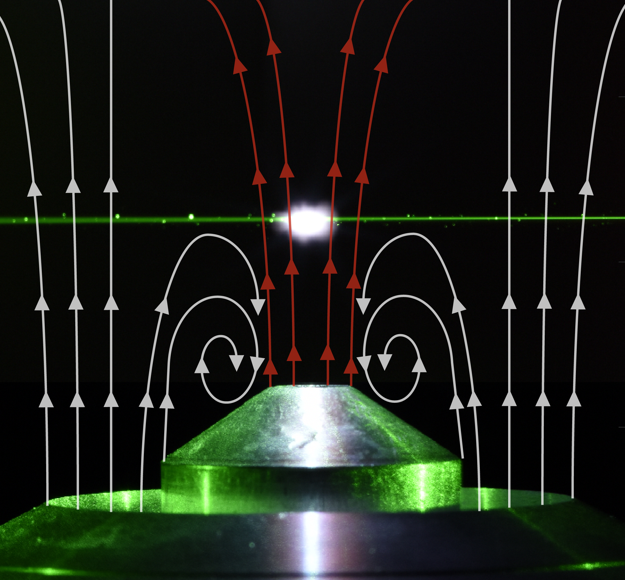
In diesem Teilprojekt wird eine Diagnosemethode entwickelt, die eine zeit- und ortsaufgelöste in-situ-Analyse der Elementzusammensetzung von Eisen- und Eisenoxidpartikeln in Strömungsreaktoren ermöglicht. Aufgrund ihrer hohen Empfindlichkeit wird die laserinduzierte Plasmaspektroskopie (LIBS) eingesetzt, um detaillierte Informationen über die Elementzusammensetzung zu erhalten. Bei LIBS wird ein gepulster Laserstrahl auf die Partikeloberfläche fokussiert und führt zur Absorption des Laserlichts und zum anschließenden Abtrag einer geringen Menge Materie, die z. B. von der Energie oder Dauer des Laserpulses abhängt. Um das Teilchen herum bildet sich ein Hochtemperaturplasma und in der frühen Phase ist breitbandiges Licht zu beobachten, während zu späteren Zeitpunkten diskrete Atomlinien emittiert werden, wenn Atome und Ionen relaxieren.
Die experimentellen Parameter, die Single-Shot-Experimente zur Partikeldiagnostik ermöglichen, müssen identifiziert und ein Schema zur Datenauswertung entwickelt werden. Die quantitative Analyse von Aerosolpartikeln mittels LIBS erfordert eine Analyse der relevanten physikalischen Prozesse, nämlich des Wärme- und Massentransfers während der Ablation, die die Dissoziation, Verdampfung, Ionisation und letztlich die atomare Anregung der Analytspezies steuern. Dieselben Prozesse spielen eine Schlüsselrolle bei partikelbezogenen Matrixeffekten, die die quantitative Analyse beeinflussen. Laser mit kürzerer Pulslänge verursachen weniger Kontinuumsemission und eine ausgeprägtere atomare Emission aufgrund eines geringeren Elektronen-Avalancheprozesses und eines kühleren Plasmas. Daher wird die Anregung eines Pikosekunden-Nd:YAG-Lasersystem mit dem üblicherweise verwendeten Nanosekunden-Anregungsschema verglichen. Mittlerweile wurden Partikel bis zu 7-10 µm vollständig ionisiert und anschließend mit LIBS analysiert. Für größere Partikel im Bereich von 30 µm, wie sie in Clean Circles relevant sind, ist die Ionisierung möglicherweise nur für einen Bruchteil des Partikels möglich. Daher muss die Elementaranalyse von großen Partikeln in parametrischen Untersuchungen erforscht werden. Eine Strömungskonfiguration, die diese parametrischen Untersuchungen an einzelnen Partikeln ermöglicht, wird entwickelt, getestet und erforscht.
Arbeitspaket Dr.-Ing. Sandra Hartl
Skalenreduzierte Modellierung für Reduktion und Oxidation von Eisen(oxid) unter Berücksichtigung heterogener Kinetik und komplexer Mischprozesse
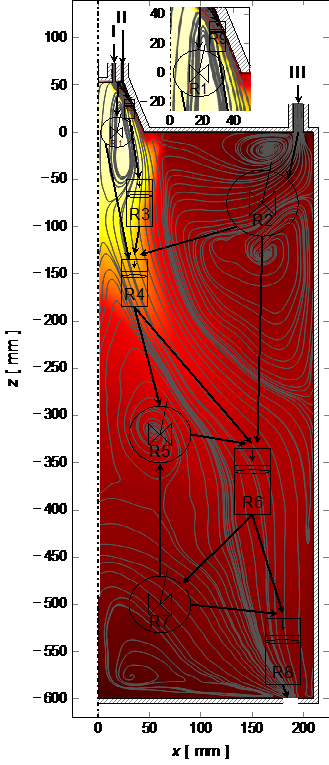
In diesem Teilprojekt werden skalen- und komplexitätsreduzierte Ansätze (z. B. Kompartmentmodelle, Reaktornetzwerk, Zonenmodell, Netzwerkmodell) zur Entwicklung eines Gesamtmodell, das die Reduktion und Oxidation von Eisen(oxid) beschreibt, entwickelt. In einem Kompartmentmodell kann das Reaktorvolumen in funktionale Kompartments mit repräsentativen Konzentrationen und Temperaturen unterteilt werden. Die Kombination idealer Reaktoren (Strömungsrohr, Rührkessel) kann folgend zur Annäherung, z. B. an das Verweilzeit- und Umsatzverhalten, realer Prozesse genutzt werden. Die Kopplung der einzelnen Reaktorelemente erfolgt über den Austausch von u. a. Spezies und Energie über Kompartmentgrenzen hinweg.
Die wissenschaftlichen Herausforderungen sind die Erweiterung, Kalibrierung und Anwendung von Kompartmentmodellen auf die Reduktion und Oxidation von Eisen(oxiden). Dazu gehört die Erweiterung der Reaktornetzwerkansätze im Hinblick auf heterogene Kinetik von Eisen(oxiden), Heizrateneffekte und den Transport von Partikeln über die Kompartmentgrenzen hinweg. Weiterhin ist eine detaillierte Analyse lokaler und globaler Verweilzeiten, Sensitivitäten und Gültigkeitsbereichen für eine Konsolidierung in einem Gesamtmodell unerlässlich. Unter Verwendung von CFD-Daten, einer (teilweise) automatisierten Aufteilung in Kompartiments, einer Erweiterung um heterogene Kinetik für Eisen(oxid) und einem detaillierten Verständnis der lokalen Verweilzeiten kann eine neue Qualität von Kompartmentmodellen erreicht werden. Diese können folgend in einem Gesamtmodell zusammengefasst und auf Anwendungen im technischen Maßstab angewendet werden.
Projekt Informationen
|
Antragsteller: |
TU Darmstadt (TUDa) Hochschule Darmstadt (hda) |
|
Koordinator: |
Prof. Dr.-Ing. Dirk Geyer |
|
Förderungsdauer: |
04/2021 - 03/2025 |
